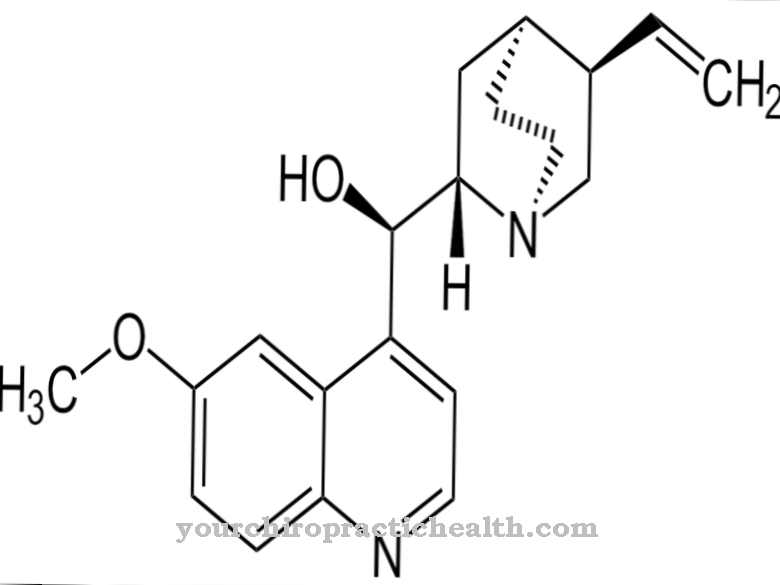
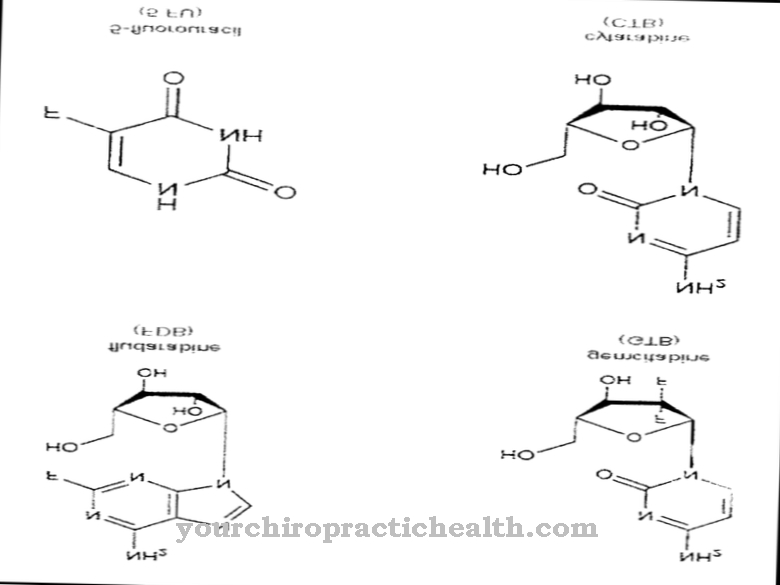
Quinolones represent a separate class of antibiotics. They all have the same basic chemical structure, which is derived from quinoline. A large number of different quinolones can be produced by exchanging the substituents on the nitrogen-containing ring system.
What are quinolones?

As a basic structure, quinolones have a nitrogen-containing ring system that is derived from quinoline. The ring system is composed of a benzene ring and a pyridine ring. In the quinolones there are still one carboxylic acid and one carbonyl group on the nitrogen-containing pyridine ring. Different substituents are bonded to the benzene ring and to the nitrogen atom of the pyridine ring.
Basically, the quinolones work by inhibiting the bacterial enzyme gyrase. The fluoroquinolones also inhibit bacterial growth in another biochemical way, which, however, has not yet been fully understood. Because of the extended effects of fluoroquinolones, they have a wide range of uses against many types of bacteria.
The active ingredient nalidixic acid, which is no longer used, serves as the starting substance for the synthesis of quinolones. Nalidixic acid, in turn, is also produced synthetically. As an alternative, the non-fluorine-containing quinolones have a piperazine ring at the corresponding point.
The spectrum of activity of the quinolones is very broad. There are representatives of this class of active ingredients that have a limited effect as well as broad and intensive effects. The basic mechanism of the antibacterial effect is the same for all quinolones. Only the substituents determine the intensity and selectivity of their influence.
Pharmacological effect
The antibacterial effect of the quinolones is based on the inhibition of the bacterial gyrase. Gyrase is an enzyme that only occurs in bacteria. It ensures the de-spiralization and restoration of the spiral structure of the DNA. The genetic code of the bacterial DNA can only be read and translated into proteins in the de-spiralized state.
However, if the gyrase is blocked, the process of untwisting the DNA is no longer active. The DNA can no longer be read properly, with the result that bacterial growth is inhibited. Existing bacteria then slowly die off. With the fluoroquinolones there is an additional mechanism that leads to the inhibition of the growth of many bacterial strains. However, this process is not yet fully understood.
The substituents determine the strength of the effect and at the same time also which bacteria are combated. Some quinolones work mainly on gram-positive bacteria, others on gram-negative bacteria. In addition to quinolones, which have a broad spectrum of activity against many types of bacteria, there are also quinolones with a very limited spectrum of activity. Since gyrase is only found in bacteria, the quinolones can only treat bacterial infections. They have no effect against fungi and viruses.
Medical application & use
Quinolones are used to fight bacterial infections. In order to be able to determine which antibiotics of this class of active substances promise the best chances of recovery, the type of bacteria must first be determined. Because not every quinolone is effective against every bacterium. The different areas of application result from this fact.
The pharmaceuticals can be administered in various ways. They can be taken orally as tablets, capsules or in the form of juices or injected as an infusion solution. All quinolones work against urinary tract and kidney infections. The drugs norfloxacin or pipemidic acid can be used for urinary tract infections. Norfloxacin is also effective against the sexually transmitted disease gonorrhea (gonorrhea). Medicines such as enoxacin can also be used for respiratory, lung or skin infections.
The drug levofloxacin has an even wider range of applications. In addition to urinary tract and respiratory infections, it can also be used to treat infections of the skin, soft tissues and eyes. The two drugs ofloxacin and ciprofloxacin cover the largest range of applications. In addition to kidney, urinary tract, respiratory tract or skin infections, the applications include bone and joint infections, infections of the digestive tract, the gall bladder, the ear, nose and throat area and the eyes. In addition, Ciprofloxacin can also be used for sepsis (blood poisoning), anthrax or cystic fibrosis (cystic fibrosis) in children.
Fluoroquinolones are very well absorbed by the body's tissues, so they can be found quickly everywhere in the body. Therefore, fluoroquinolones can also be used to fight infections that are difficult to achieve with other drugs, such as bone or prostate infections.
Risks & side effects
Quinolones interact with other drugs that either reduce their effectiveness or lead to side effects. The effectiveness of quinolones is reduced by divalent calcium or magnesium salts as well as by antacids, since they form complexes together.
When used with anti-rheumatic agents and steroids, states of excitement can occur. Furthermore, different side effects can occur when using the medication. Nausea and vomiting are observed. Diarrhea and abdominal pain are less common.
Neurological disorders such as increased excitability, restlessness, insomnia, dizziness or even hallucinations can occur in rare cases.
There is an increased sensitivity to solar radiation during treatment with quinolones. Sunbathing or sunbathing is contraindicated during quinolone therapy. Studies also indicate that quinolones have properties that damage cartilage. Cardiac arrhythmias are also possible due to disturbances in the conduction of excitation.
Very rare side effects are changes in the blood count and allergic reactions. Quinolones should not be administered during pregnancy or breastfeeding. The same applies to use in children and adolescents. A contraindication is also present in the case of cerebral cramps, severe renal insufficiency or cardiac arrhythmias.














.jpg)

.jpg)