Propiverine belongs to the active ingredient groups of antispasmodics and muscle relaxants. Preferably, this drug is prescribed to children with urinary incontinence.
What is propiverine?

Propiverine is also under the alternative name Propiverinum known. This drug is assigned to the active ingredient groups of antispasmodics and muscle relaxants. Propiverine is the drug of choice for children with urinary incontinence caused by increased tension in the urinary muscles. This medicine is also used in patients with overactive bladder due to paraplegia, multiple sclerosis and spinal cord damage.
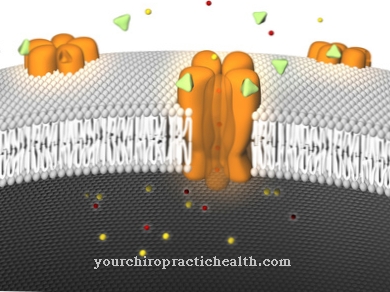
Propiverine has active metabolites that dock on the muscarinic receptors (anticholinergic system) and thus cause the bladder to relax. In vitro, there is also a calcium antagonistic effect on the cells involved in the smooth muscles of the urinary bladder.
Pharmacological effect
Propiverine is preferably used in children with urinary incontinence. This incontinence is due to an over-excitability of the urinary muscle, which causes deviating urinary behavior, such as nocturnal wetting, nocturnal urination (nocturia), sudden loss of urine and the leakage of small amounts of urine (pollakiuria).
Frequently, uncontrolled urinary behavior occurs in patients with paraplegia, multiple sclerosis and spinal cord damage due to an overactive urinary bladder. In these cases, too, propiverine is the drug of choice.
A third area of application is the treatment of bladder damage after surgery and radiation. As a spasmolytic, propiverine has a direct effect on the activity of muscles and nerves. In the area of the muscles, the drug is active on the muscle fibers of the hollow organs, which include the bladder.
In the area of the parasympathetic nerve fibers, which belong to the autonomic nervous system, propiverine with its anticholinergic effect displaces the body's own messenger substance acetylcholine, which is attached to the nerve fibers, and prevents excessive excitation of these receptors, which leads to the unwanted reflex of excessive urination. The urge to urinate is noticeably reduced and the urinary bladder has a greater capacity.
Medical application & use
Propiverine has a long half-life of 14 to 20 hours. Each film-coated tablet contains 15 mg propiverine hydrochloride. Taking one film-coated tablet twice a day is usually sufficient. Due to the favorable profile of action, patients with minor symptoms get along with one tablet a day. In the case of nerve-related disorders of urination, the daily dose can be increased to three tablets, which corresponds to a maximum daily dose of 45 mg propiverine hydrochloride.
Patients with impaired renal function should not exceed a maximum daily dose of 30 mg. In children, treatment for an overactive bladder should not begin before the age of five, as organ development is not yet complete at this age. Even if propiverine is the drug of choice for children and is also considered to be well tolerated, treatment should only take place within the framework of an overall therapeutic concept (urotherapy). Overall, a low body weight of less than 35 kg is not suitable for the administration of film-coated tablets containing 15 mg of this active ingredient. In this case, film-coated tablets with a low propiverine content of 5 mg are prescribed.
Dry mouth is described as the most common accompanying symptom when taking anticholinergic drugs. In contrast to oxybutynin, another active ingredient for treatment in the drug therapy of incontinence, propiverine is better tolerated. Medical professionals believe that the rate of anticholinergic side effects is reduced due to the drug's dual action.
You can find your medication here
➔ Medicines for bladder and urinary tract healthRisks & side effects
Propiverine also has risks and side effects that may be a contraindication. The drug must not be used in the case of known hypersensitivity to the active ingredient. Patients with bowel obstruction, inadequate bowel movement (bowel atony), inflammatory bowel disease, enlarged colon due to bacterial colonization (toxic megacolon) and myasthenia gravis must not take propiverine. Other contraindications are liver dysfunction and glaucoma (narrow-angle glaucoma).
A careful risk-benefit analysis must be carried out in patients with kidney and nerve dysfunction. In patients with a narrow anterior chamber angle, the drug can trigger a glaucoma attack due to its pupil-expanding effect. Side effects can include various heart problems, heartburn, inflammation of the esophagus and benign enlargement of the prostate.
Many patients suffer from visual disturbances, abdominal pain, digestive problems, decreased visual acuity, fatigue and tiredness. Occasionally lower blood pressure, tremors, dizziness, nausea, drowsiness, urinary retention and taste disturbances are possible.
Nocturnal and frequent need to urinate as a result of kidney disease, organic bladder disease, bladder cancer and heart muscle weakness are clinical pictures that are not suitable for treatment with propiverine. During pregnancy and breastfeeding, this active ingredient is only prescribed after a strict risk-benefit analysis. Since the active ingredient is suitable for children, there are no fundamental contraindications. However, it is important to adjust the dose to the body weight of the little patient.
With simultaneous intake of various psychotropic drugs such as pediatric antidepressants, benzodiazepines and neuroleptics, the effect is increased. This also applies to the simultaneous use of muscle relaxing drugs such as muscarinic receptor antagonists. Patients undergoing treatment for cardiac arrhythmias, Parkinson's and asthma may only take propiverine after prior consultation with the attending physician.
The combination of propiverine and metoclopramide for gastrointestinal problems reduces the effectiveness of both drugs. Isoniazid, used to treat tuberculosis, may cause a drop in blood pressure.
The use of propiverine can also impair the ability to react, especially when taking central depressant drugs such as psychotropic drugs, to such an extent that using machines and driving a car is not safe. Sometimes drugs can cause allergic reactions.







.jpg)

.jpg)


.jpg)