At Pyrazinamide is a drug used to treat tuberculosis (tuberculostatic). The substance has been used since the 1950s to combat lung disease as part of a combination therapy.
What is pyrazinamide?
Pyrazinamide (PZA for short) is an antibiotic that has been used since the 1950s to fight tuberculosis. The remedy is also often called Pyrazine carboxamide is water-soluble and white. Due to its intended use as a tuberculosis drug, it is classified as a tuberculostatics.
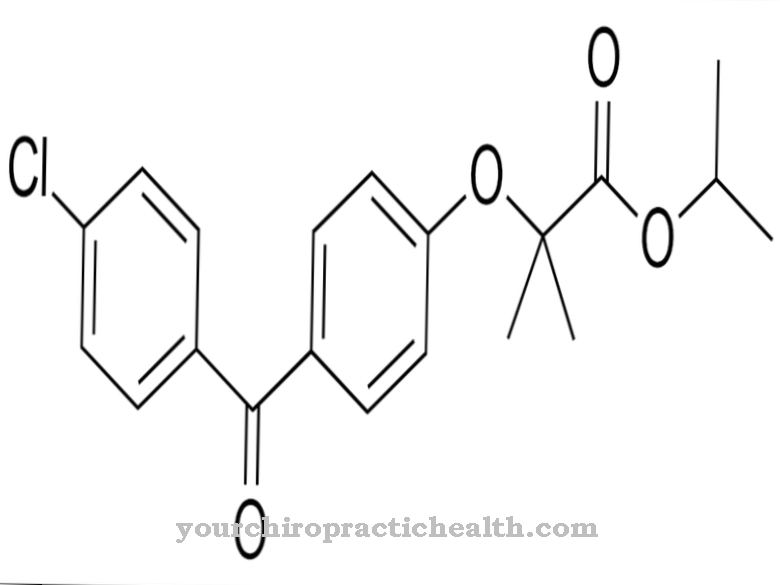
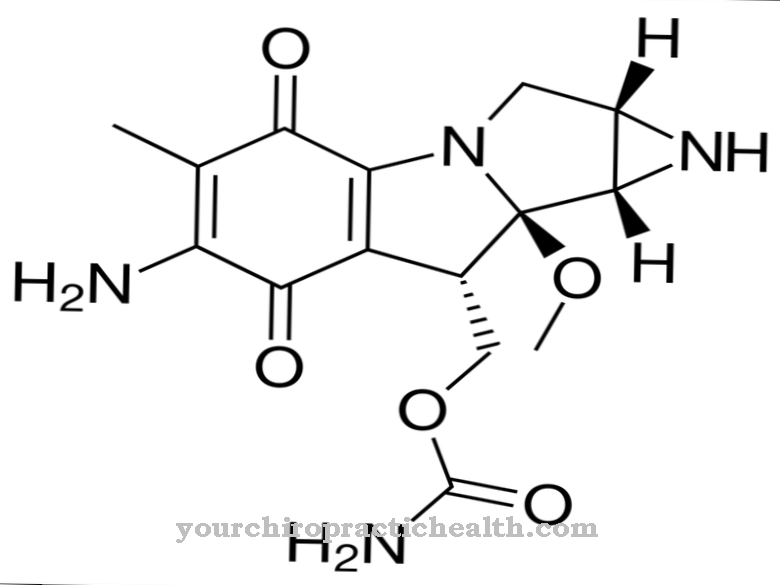
In chemistry, the empirical formula C5-H5-N3O is used to denote the substance. The moral mass of pyrazinamide is 123.11 g • mol − 1. The active ingredient pyrazinamide is u. a. sold under the trade names Pyrafat®, Rifater®, Rimstar® and Tebesium Trio®.
PZA only acts on the tuberculosis pathogen (Mycobacterium tuberculosis). There is thus no effectiveness in relation to the bovine form of the bacterium (Mycobacterium bovis) or forms that differ from the basic type (atypical mycobacteria).
Pharmacological effect
Although PZA has been used to combat tuberculosis since the 1950s, the exact mechanism of action of the substance had not been clarified for a long time. Only the effectiveness of the treatment was trusted. The mechanism of action of pyrazinamide was only fully elucidated in 2011. While conventional antibiotics primarily kill bacteria that are still growing, PZA primarily kills growing bacteria. Thus, its mechanism of action represents a deviation from the norm. Because growing bacteria (so-called persisters) are usually far less sensitive to antibiotics than those that are already in a state of rest.
Pyrazinamide only works in the body. No effect could be found in test tubes, which contributed to the fact that the mechanism of action remained unknown for so long. Even before 2011, however, it was known that PZA works as a prodrug. The substance converts pyrazinoid acid in the body and works in an acidic environment. Pyranicide binds to a cell protein (RspA S1) and thus prevents trans-translation in the tuberculosis bacterium. As a result, it is no longer possible for them to protect themselves against toxic protein fragments.
These fragments are made by the bacterium itself when it is under stress. Because of this mechanism of action, PZA shortens the treatment of tuberculosis from 9 to 12 months to usually 6 months.
Medical application & use
Pyrazinamide has been used to fight the lung disease tuberculosis since the 1950s. The substance is one of the antibiotics and is a tuberculostatic drug. There is only one indication for tuberculosis in humans.
Pyrazinamide is not applicable to atypical forms of the bacterium (atypical mycobacteria) or the bovine form of bacteria (MYcobacterium bovis). When taken early, it shortens the average duration of treatment for tuberculosis from around 9 to 12 months to 6 months.
The drug is usually used in combination therapy. Patients also often take isoniazid, ethambutol, and rifampicin as part of their therapy. However, the exact combination can vary depending on the type of therapy chosen. The form of therapy depends on the duration and severity of the disease and the age of the patient. The average duration of treatment also depends on these factors.
You can find your medication here
➔ Medicines against coughs and coldsRisks & side effects
Pyrazinamide can cause side effects. However, this is not necessarily the case. Before taking it for the first time, it should be checked whether there is any intolerance to pyrazinamide or related substances. In the event of an allergy, use or ingestion is prohibited.
In addition, pyrazinamide is contraindicated in severe liver disorders, kidney dysfunction, acute gout attacks and during pregnancy and breastfeeding. Caution should also be exercised in patients who regularly consume large amounts of alcohol.
Since pyrazinamide can affect the liver and kidney function, the function of the organs should be monitored at regular intervals during therapy. This check should continue for a few weeks after treatment.
The following side effects are also known: Common (less than 1 in 10 patients, but more than 1 in 100): loss of appetite, nausea, nausea, weight loss, liver damage, heartburn, sensitivity to light and an increase in the level of uric acid in the blood.
Rare side effects include headache, excitability, insomnia, and dizziness. Rare side effects are those that affect less than 1 in 1,000 people, but more than 1 in 10,000.
In less than 1 in 10,000 cases, disorders of the blood formation system (e.g. anemia) and thrombocytopenia (very rare) occurred. The instructions of a doctor and a pharmacist must be followed. You should be contacted immediately if these and other side effects occur.





.jpg)

.jpg)


.jpg)