It is an everyday phenomenon that when entering a dark room, the initially poor eyesight improves as the eyes adapt to the light conditions. This is known as dark adaptation and is essential for that Scotopic vision at night.
What is scotopic vision?

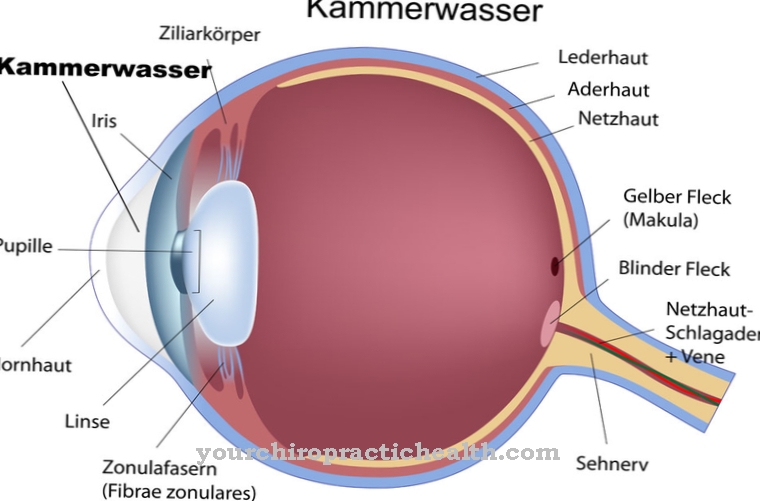
Scotopic vision refers to seeing in the dark. In contrast to photopic vision, it is realized by the rod sensory cells of the retina, because they are particularly suitable for light-dark vision due to their increased sensitivity to light.
If the rods are damaged due to inherited or acquired changes, this can lead to a severe reduction in vision in the dark, known as night blindness.
Function & task
There are two different types of photoreceptors on the retina of the human eye that are needed for the process of vision: rods and cones. The cones are responsible for color vision in brightness, also known as photopic vision. The rods take over the visual process in low light or at night, i.e. scotopic vision.
The fact that the rod sensory cells of the eye cannot differentiate between different colors is also the reason for our limited color perception in the dark. However, rods and cones are not evenly distributed over the retina. The highest density of sensory cells and thus the sharpest image resolution is achieved at the so-called yellow spot, the fovea centralis. However, there are only cones there that are of little use when looking at night. Scotopic vision is therefore optimal when the eye is aligned in such a way that the image on the retina is not created on the yellow spot, but next to it (parafoveal).
In principle, both types of sensory cells convert light into a signal to the brain using the same mechanism. The energy of the incident light leads to a structural change in a protein, the rhodopsin. This triggers a signal cascade in the cell, as a result of which less glutamate is released. Downstream nerve cells register this and transmit an electrical signal to the brain.
During the transition to seeing in the dark, for example when entering a dark room, there is a dark adaptation that consists of four effects. A quick aspect is the pupillary reflex. When there is little light exposure, the pupil is set wide so that as much light as possible can fall through the opening of the iris onto the retina. In addition, the light sensitivity of the photoreceptors is increased. Your stimulus threshold drops, among other things, due to an increased concentration of rhodopsin, which is only possible in the dark.
On the other hand, in the dark there is a switch from cone to rod vision, since the rods per se already have a higher light sensitivity than the cones. This transition takes a certain amount of time and is also known as the Kohlrausch kink.
Finally, with increasing darkness, the lateral inhibition in the retina decreases and thus the size of the receptive fields increases. The result is a stronger convergence of the signals on the downstream ganglion cells, which are responsible for the transmission to the brain and are thus more strongly aroused. However, this increased convergence takes place at the expense of the resolving power, i.e. the visual acuity.
You can find your medication here
➔ Medicines for eye infectionsIllnesses & ailments
A defect or weakening of scotopic vision is called night blindness. In this case, the eye can no longer (adequately) adapt to the dark, and vision in twilight or in the dark is reduced or no longer available. This disorder can be inherited (congenital) or acquired. However, night blindness can also occur as an accompanying symptom in other diseases.
Congenital night blindness can be triggered, for example, by hereditary mutations in proteins that are important for the visual process, such as the S-arrestin in Oguchi syndrome. Retinitis pigmentosa, a group of hereditary retinal diseases for which causal mutations in over 50 different genes are currently known, is also genetic. The onset of this disease, which is usually noticeable for the first time in childhood, adolescence or in young adults, is often indicated by night blindness. In addition to restricted scotopic vision, a loss of visual field, increased sensitivity to glare and an increasing loss of color vision often occur in the course of retinits pigmentosa.
Even with a cataract (cataract) symptoms occur that are described by the patients as night blindness. However, the cause here is not a malfunction of the rods in the retina, but rather a clouding of the lens.
Similarly, in the course of illness with diabetes mellitus, scotopic vision can be restricted, which is known as diabetic retinopathy. In the case of liver amaurosis, in addition to night blindness, patients often have increased sensitivity to glare, nystagmus (involuntary eye tremors) and generally reduced eyesight.
A distinction between these forms of night blindness is that caused by a lack of vitamin A. Vitamin A is necessary for the body's own production of the visual pigment rhodopsin. An improvement in this form of night blindness can therefore be achieved by administering vitamin A. In the western industrialized nations, however, night blindness caused by deficiency is very rare, as the need for vitamin A can easily be met with a balanced diet.
In the case of certain risk factors for a vitamin A deficiency such as various intestinal diseases, inflammation of the pancreas, eating disorders or pregnancy, however, particular attention should be paid to an adequate supply of vitamin A. In developing countries, vitamin A deficiency due to malnutrition is still a cause of dramatic blindness rates in children.

.jpg)