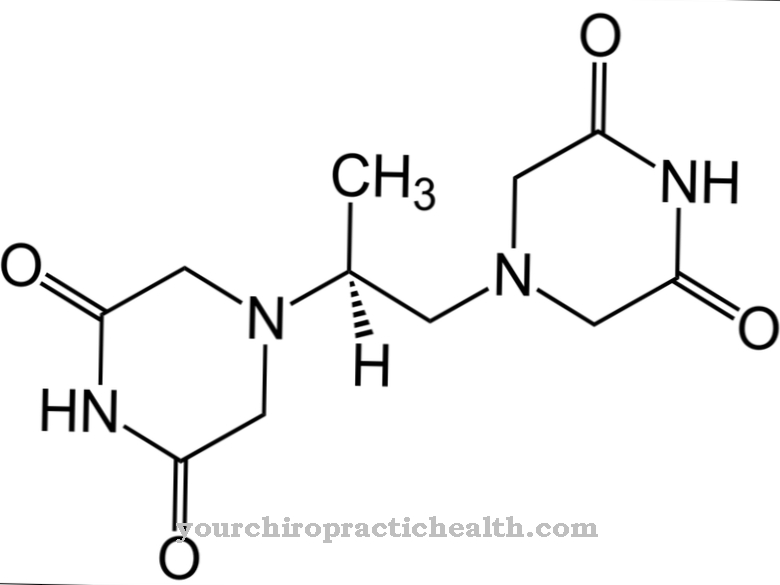
Droperidol is a drug from the class of neuroleptics. It is given as a preventative measure against nausea and vomiting after surgery.
What is droperidol?

The drug droperidol belongs to the group of butyrophenones. Butyrophenones are a group of drugs that are mainly used in the pharmacological therapy of schizophrenia. Droperidol also has antipsychotic properties. In combination with the antiemetic effect, Droperidol is therefore suitable as a remedy for postoperative nausea and as a sedative in neuroanesthesia.
The drug is a derivative of benperidol. Droperidol comes in a white powder form at room temperature. The powder is hardly soluble in water. The drug is commercially available as a solution for injection. It has been approved in Switzerland since 2006. After it was withdrawn from the market in 2001, the drug was approved again in Germany in 2008.
Pharmacological effect
Like most neuroleptics, Droperidol also has a binding affinity for D2 receptors in the central nervous system. D2 receptors are also called dopamine receptors. They serve as a docking point for dopamine, a neurotransmitter. Dopamine has an inhibitory effect on the extrapyramidal motor system via the D2 receptors.
Droperidol mainly affects the D2 receptors in the area postrema. The area postrema is located in the brain stem and together with the nucleus tractus solitarii forms the vomiting center. The transmitter dopamine plays an important role in vomiting. Dopamine antagonists like droperidol block the D2 receptors and thus inhibit nausea.
Droperidol also has a low affinity for D3 receptors. These receptors also serve as a docking point for dopamine. D3 receptors are mainly found in the limbic system and in the cortical areas of the brain. They play a role in emotional and cognitive processes. The inhibition of the D3 receptors leads to an alleviation of psychotic symptoms.
Droperidol can also bind to 5-HT2 receptors. The inhibition of the responsiveness of the receptors has, inter alia, an anxiolytic effect.
Medical application & use
Droperidol was administered together with the drug Thalamonal prior to surgery until the 1980s. The combination of the active ingredients fentanyl] and droperidol should sedate the patient.At the same time, they should be relieved of their fear of the operation. Many of the patients, however, complained of depression, panic and agitation with simultaneous fatigue. Therefore the drug was only used by anesthetists in exceptional cases. Benzodiazepines are now more commonly used for this purpose.
In 2001 the oral dosage form of Droperidol was withdrawn from the market. With high-dose long-term therapy, side effects occurred that impaired the heart. With the peroral application, the parenteral application form was also withdrawn from the market. The drug was only approved again in Germany in 2008.
Today it is available in anesthesia for the prophylaxis and therapy of nausea and vomiting after operations. It can be used in adults and in children over two years of age. Droperidol can also be administered to prevent nausea and vomiting caused by opioids in patient-controlled analgesia. Patient-controlled analgesia allows the patient to self-administer a pain reliever. Classically, an opioid is administered intravenously using a pain pump.
You can find your medication here
➔ Medicines against nausea and vomitingRisks & side effects
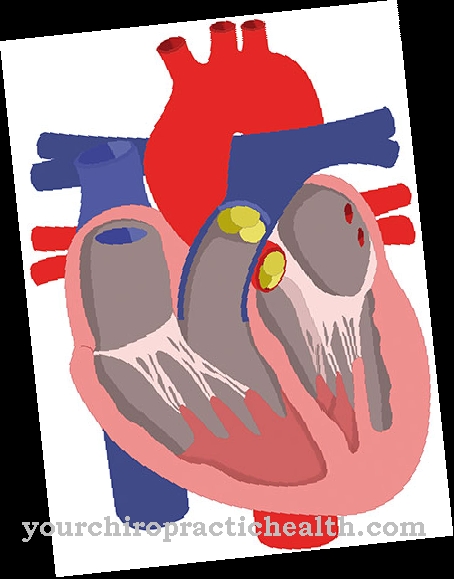
Droperidol must not be used in patients who are known to be hypersensitive or allergic to droperidol or to any of the ingredients in the drug. Hypersensitivity or allergies to butyrophenones are also contraindications. Droperidol must not be administered if there is a known or suspected prolonged QT time in the ECG. In women, the QT time must not exceed 440 ms, men must not exceed 450 ms. This limitation also applies to patients who have inherited prolonged QT times in their families and to patients who are taking drugs that have been shown to prolong QT time.
Further contraindications for the use of Droperidol are potassium deficiency and magnesium deficiency. Bradycardia, i.e. a slow heartbeat, is also a contraindication. Another drug should also be used in patients with pheochromocytoma. Exclusion criteria are also comatose states, Parkinson's disease and severe depression.
Depressive episodes may occur when taking Droperidol. Some patients also complain of nervousness, impaired memory and confusion.





.jpg)

.jpg)


.jpg)