Nucleobases are the building blocks from which the long chains of DNA and RNA molecules are composed in their phosphorylated nucleotide form.
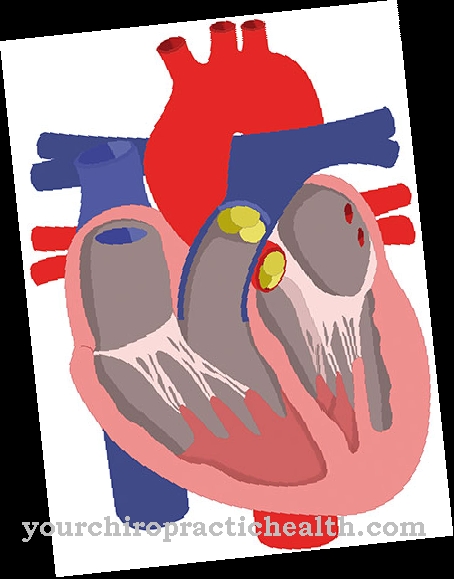
In the DNA, which forms double strands similar to ropes, the 4 occurring nucleic bases form solid pairings with the respective complementary base via hydrogen bridges. The nucleobases consist of either a bicyclic purine or a monocyclic pyrimidine skeleton.
What are nucleobases?
The 4 nucleobases adenine, guanine, cytosine and thymine, as building blocks of the long DNA double helix molecule chains, form the constant pairings adenine-thymine (A-T) and guanine-cytosine (G-C).
The two bases adenine and guanine each consist of a modified bicyclic six and five ring of the purine basic structure and are therefore also referred to as purine bases. The basic structure of the other two nucleic bases, cytosine and thymine, consist of a heterocyclic aromatic six-membered ring, which corresponds to a modified pyrimidine skeleton, which is why they are also referred to as pyrimidine bases. Since the RNA is mostly present as single strands, there are initially no base pairs. This only takes place during replication via the mRNA (messenger RNA).
The copy of the RNA strand consists of the complementary nucleobases analogous to the second strand of DNA. The only difference is that uracil is substituted for thymine in RNA. The DNA and RNA chain molecules are not formed in their pure form by the nucleobases, but instead, in the case of DNA, combine with the 5-sugar deoxyribose to form the corresponding nucleoside. In the case of RNA, the sugar group consists of ribose. In addition, the nucleosides are phosphorylated with a phosphate residue to form so-called nucleotides.
The purine bases hypoxanthine and xanthine, which are also found in DNA and RNA, correspond to modified thymine. Hypoxanthine is formed from adenine by replacing the amino group (-NH3) with a hydroxyl group (-OH), and xanthine is formed from guanine. Both nucleobases do not contribute to the transmission of genetic information.
Function, effect & tasks
One of the most important functions of the nucleobases that make up the double strands of DNA is to show presence at the intended position.
The sequence of the nucleobases corresponds to the genetic code and defines the type and sequence of amino acids from which proteins are composed. This means that the most important function of the nucleobases as a component of DNA consists of a passive, static role, i.e. they do not actively intervene in the metabolism and their biochemical structure is not changed by messenger RNA (mRNA) during the reading process. That partly explains the longevity of DNA.
The half-life of mitochondrial DNA (mtDNA), during which half of the originally existing bonds between the nucleobases disintegrates, depends heavily on the environmental conditions and varies between around 520 years under average conditions with positive temperatures and up to 150,000 years under permafrost conditions .
As part of the RNA, the nucleobases play a somewhat more active role. In principle, when cells divide, the DNA double strands are broken up and separated from one another in order to be able to form a complementary strand, the mRNA, which, so to speak, forms the working copy of the genetic material and serves as the basis for the selection and sequence of the amino acids from which the intended proteins are assembled. Another nucleic base, dihydrouracil, occurs only in the so-called transport RNA (tRNA), which is used to transport amino acids during protein synthesis.
Some nucleobases fulfill a completely different function as part of enzymes that actively catalytically enable and control certain biochemical processes. Adenine fulfills its best-known task as a nucleotide in the energy balance of cells. Adenine plays an important role as an electron donor as adenosine diphosphate (ADP) and adenosine triphosphate (ATP) as well as a component of the nicotinamide adenine dinucleotide (NAD).
Education, occurrence, properties & optimal values
In the non-phosphorylated form, nucleobases consist exclusively of carbon, hydrogen and oxygen, substances that are ubiquitous and freely available. The body is therefore able to synthesize nucleobases itself, but the process is complex and energy-consuming.
Therefore, the recovery of nucleic acids by recycling is preferred, e.g. B. through the breakdown of proteins that contain certain compounds that can be isolated and converted into nucleic acids with little energy expenditure or even with energy gain. Nucleic acids do not usually occur in pure form in the body, but mostly as nucleosides or deoxynucleosides with an attached ribose or deoxyribose molecule. As a component of DNA and RNA and as a component of certain enzymes, the nucleic acids or their nucleosides are also reversibly phosphorylated with one to three phosphate groups (PO4-).
There is no reference value for an optimal supply of nucleobases. A deficiency or an excess of nucleobases can only be determined indirectly via certain disorders in the metabolism.
Diseases & Disorders
The nature of the dangers, disturbances and risks that are brought into connection with the nucleobases are errors in the number and sequence on the DNA or RNA strands, which lead to a change in the coding for protein synthesis.
If the body cannot remedy the fault via its repair mechanisms, it comes to the synthesis of biologically inactive or usable proteins, which in turn can lead to mild to serious metabolic disorders. It can e.g. B. gene mutations are present that can trigger symptomatic diseases from the outset via metabolic disorders, which may be incurable. But even in a healthy genome, copying errors can occur during the replication of the DNA and RNA chains, which affect the metabolism.
A known metabolic disorder in the purine balance is z. B. back to a genetic defect on the x chromosome. Because of the genetic defect, the purine bases hypoxanthine and guanine cannot be recycled, which ultimately promotes the formation of urinary stones and gout in the joints.







.jpg)

.jpg)


.jpg)