In a biosynthesis, the inactive protein prothrombin becomes the coagulation factor Thrombin educated. Thrombines convert fibrinogens into fibrin and thus realize the last step in the coagulation cascade. In the case of genetic prothrombin mutations, the increased prothrombin concentration in the plasma leads to a tendency to thrombosis.
What is thrombin
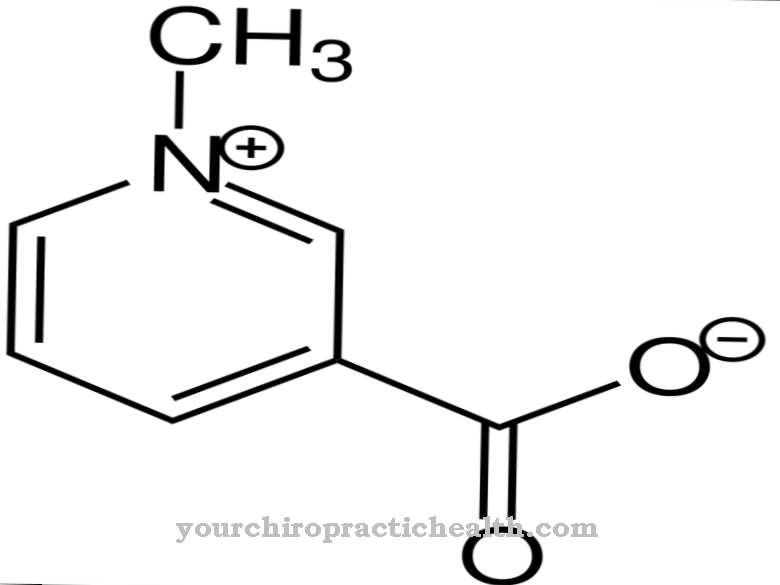
Thrombin occurs as a protein in the blood plasma and is involved in the plasmatic coagulation of the blood. Its precursor is known as factor II in blood clotting. Thrombin is produced in the liver, from where it is permanently released into the blood. So that the blood does not clot in healthy vessels, the body produces its own antithrombins, which have an inhibitory effect on clotting. With open wounds and injuries in the tissue, thrombin is formed directly at the site of the injury.
In particular, its inactive precursor prothrombin is therefore present in the plasma, whereas the actual thrombin is only found in small quantities in the plasma. The enzyme was first described by Schmidt, who mentioned it in his book on blood theory at the end of the 19th century. With heparin and similar substances, the pharmaceutical industry has developed numerous agents for inhibiting thrombin, which reduce blood clotting during medical procedures such as dialysis. These antithrombines are modeled on the body's own antithrombines.
Anatomy & structure
The body's own thrombin is a protein. It is formed in the liver as part of a biosynthesis from the inactive form prothrombin. For humans, on the genetic basis, the F2 gene on chromosome 11 plays a role in this process and the development of prothrombin. This gene spans more than 20,000 base pairs and makes up 14 exons. The mRNA, which reaches 1,997 bases, is translated into protein of 622 amino acids after transcription.
Via modifications, the product of this translation results in prothrombin and thus the precursor of thrombin, which comprises 579 amino acids. This thrombin precursor is inactive until it is converted to thrombin in the biosynthesis. This conversion takes place through the enzymatic cleavage of the prothrombin. The enzymatic prothrombinase complex plays the main role in this process. The conversion of inactive prothrombins to active thrombins takes place in a vitamin K-dependent manner and is a step in the so-called coagulation cascade.
Function & tasks
Thrombin catalyzes the last step in the coagulation cascade. This cascade protects the body from major blood loss and closes the wound to protect against infection. The coagulation cascade is the systematic activation of the individual coagulation factors. As a so-called serine protease, thrombin initiates the conversion of fibrinogen into fibrin. To do this, thrombin hydrolyzes a so-called arginylglycine bond in the α and β chains of the fibrinogens and splits off four polypeptides.
The molecular weight of 340,000 for fibrinogens is reduced to around 270,000 Daltons for fibrin. Polymerization takes place through the fibrin. In the process, non-covalent bonds are formed. Due to the coagulation factor XIII, the covalent peptide bonds are finally formed from these bonds and the coagulation is complete. To put it simply, the thrombin splits off the fibrinogen for each peptide residue. The conversion takes place proteolytically, i.e. through the breakdown of proteins. During this process, fibrin threads are formed from the fibrinogen.
The blood changes its consistency in this way. Instead of being in liquid form, the threads present it in the form of jelly, which is finally processed by coagulation factor XIII into a network of fibrins. Fibrinogens are also referred to as coagulation factor I because of their relevance in the coagulation cascade. In this system, thrombin serves as its catalyst and, so to speak, takes on coagulation functions behind the scenes by helping to change the blood consistency in the event of an injury.
You can find your medication here
➔ Medicines for wound treatment and injuriesDiseases
One of the most important diseases with involvement of thrombin is the so-called prothrombin mutation or factor II mutation. People with these bleeding disorders are at increased risk of developing a blood clot. Compared to the blood of healthy people, their blood coagulates faster. The reason for this is a change in the genetic information of the prothrombin. This genetic abnormality is a point mutation in the prothrombin gene.
Due to the incorrect genetic information, significantly larger amounts of prothrombins are present in the patient's blood. As a result, their blood tends to clot. As a result, thromboses tend to set in or, if the thrombus is spread, even embolisms. Heart attacks and strokes or kidney attacks can be the result. In combination with risk factors such as smoking or taking the birth control pill, vascular occlusions and heart attacks are even more common for those affected by the prothrombin mutation. The mutation affects about two out of 100 people in Germany and can be detected by genetic analysis.
Treatment with antithrombin can significantly reduce the risk of serious consequences. A deficiency of prothrombin in the blood can also be congenital. With such a deficiency, those affected suffer from a tendency to bleed. In addition to hereditary coagulation disorders, there may also be an acquired bleeding tendency, for example if the liver is damaged. Even with deficiency symptoms, the coagulation is sometimes disturbed. Thrombine are produced with the consumption of vitamin K, so a deficiency in this vitamin in particular can manifest itself in an insufficiency in the coagulation cascade.



.jpg)
.jpg)





.jpg)





.jpg)