Actin is a structural protein found in all eukaryotic cells. It participates in the structure of the cytoskeleton and muscles.
What is actin
Actin is an evolutionarily very old protein molecule. As a structural protein, it is contained in the cytoplasm of every eukaryotic cell and in the sarcomere of all muscle fibers.
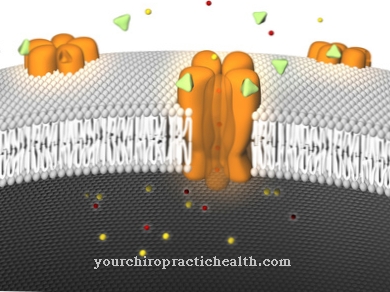
Together with the microtubules and intermediate filaments, it forms the cytoskeleton of every cell in the form of actin filaments. It is jointly responsible for the formation of the cell structure and the movement of molecules and cell organelles within the cell. The same applies to the cohesion of the cells via tight junctions or adherens junctions. In the muscle fibers, actin, together with the proteins myosin, troponin and tropomyosin, creates the muscle contractions.
Actin can be divided into the three functional units alpha-actin, beta-actin and gamma-actin. Alpha-actin is the structural component of muscle fibers, while beta and gamma-actin are mainly found in the cytoplasm of cells. Actin is a very conserved protein, which occurs in unicellular eukaryotic cells with very small deviations in the amino acid sequence. In humans, 10 percent of all protein molecules in muscle cells consist of actin. All other cells still contain 1 to 5 percent of this molecule in the cytoplasm.
Function, effect & tasks
Actin fulfills important functions in cells and muscle fibers. In the cytoplasm of the cell, as part of the cytoskeleton, it forms a dense, three-dimensional network that holds the cellular structures together.
At certain points in the network, the structures strengthen to form membrane bulges such as microvilli, synapses or pseudopodia. The Adherens Junctions and Tight Junctions are available for the cell contacts. Overall, actin contributes to the stability and shape of cells and tissues. In addition to stability, actin also ensures the transport processes within the cell. It binds important structurally related transmembrane proteins so that they remain in close proximity. With the help of myosins (motor proteins), the actin fibers also carry out transport over short distances.
For example, the vesicles can be transported to the membrane. Longer stretches are taken over by the microtubules with the help of the motor proteins kinesin and dynein. Actin also ensures cell mobility. Cells must be able to migrate within the body on many occasions. This applies in particular to immune reactions or wound healing as well as to general movements or changes in the shape of cells. The movements can be based on two different processes. On the one hand, the movement can be triggered by a directed polymerization reaction and, on the other hand, via the actin-myosin interaction.
In the actin-myosin interaction, the actin fibers are built up as fibril bundles that function like pulling ropes with the help of myosin. Actin filaments can cause cell outgrowths in the form of pseudopodia (filopodia and lamellipodia). In addition to its diverse functions within the cell, actin is of course responsible for the muscle contraction of both the skeletal muscles and the smooth muscles. These movements are also based on the actin-myosin interaction. To ensure this, many actin filaments are linked to other proteins in a very orderly manner.
Education, occurrence, properties & optimal values
As already mentioned, actin is found in all eukaryotic organisms and cells. It is an inherent part of the cytoplasm and ensures the stability of the cells, the anchoring of structurally related proteins, the short-distance transport of vesicles to the cell membrane and cell motility. Without actin, the cell would not be able to survive. There are six different actin variants, which are divided into three alpha variants, one beta variant and two gamma variants.
The alpha actins are involved in the development and contraction of the muscles. Beta-actin and gamma-1-actin are of great importance for the cytoskeleton in the cytoplasm. Gamma-2-actin, in turn, is responsible for the smooth muscles and the intestinal muscles. During the synthesis, monomeric globular actin, which is also known as G-actin, is initially formed. The individual monomeric protein molecules, in turn, polymerize to form a filamentary F-actin.
During the polymerization process, several spherical monomers combine to form a long, thread-like F-actin. Both the construction and the breakdown of the chains are very dynamic. In this way, the actin framework can be quickly adapted to current requirements. In addition, this process also ensures cell movements. These reactions can be inhibited by so-called cytoskeletal inhibitors. With these substances, either polymerizations or depolymerizations are inhibited. They are of medical importance as medicinal products in the context of chemotherapy.
Diseases & Disorders
Since actin is an essential component of all cells, many structural changes caused by mutations lead to the death of the organism. Mutations in genes for alpha actins can cause muscle disorders. This is especially true for alpha-1-actin.
Due to the fact that alpha-2-actin is responsible for the aortic muscles, a familial thoracic aortic aneurysm can occur if the ACTA2 gene is mutated. The ACTA2 gene codes for alpha-2-actin. A mutation in the ACTC1 gene for cardiac alpha actin causes dilated cardiomyopathy. Furthermore, a mutation of ACTB as a gene for cytoplasmic beta-actin can cause large-cell and diffuse B-cell lymphoma. Some autoimmune diseases can have elevated levels of actin antibodies.
This applies in particular to autoimmune liver inflammation. It is a chronic course of hepatitis, which leads to liver cirrhosis in the long term. Here an antibody against smooth muscle actin is found. In terms of differential diagnosis, autoimmune hepatitis is not so easy to differentiate from chronic viral hepatitis. Because in chronic viral hepatitis, antibodies against actin can also be stimulated to a lesser extent.






.jpg)

.jpg)


.jpg)