The Bioavailability is a measurable quantity that relates to the active substance of drugs. The value corresponds to the percentage of an active ingredient that reaches the systemic distribution in the organism in unchanged form. The bioavailability corresponds to the speed and the extent to which a drug is absorbed and can develop its effect at its destination.
What is the bioavailability?

Bioavailability is a pharmacological term that refers to the percentage of an active ingredient in a certain drug dose that is available in unchanged form to the system and bloodstream. The bioavailability thus corresponds to a measure of the speed and extent to which a certain drug is absorbed and ultimately reaches its respective site of action.
A special measure of bioavailability is that absolute bioavailability. By definition, intravenously administered drugs have 100 percent bioavailability. The absolute bioavailability is therefore the bioavailability of a drug compared to its intravenous administration. Of the relative bioavailability is always used when a form of administration of an active ingredient is compared with another form of administration.
In pharmacokinetics, bioavailability is an important parameter, especially in connection with drug approval.
Function & task
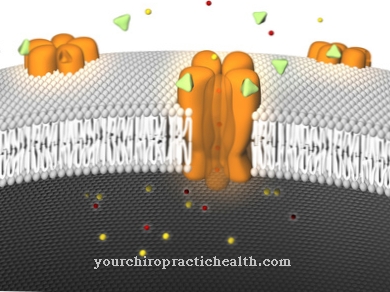
After taking a certain drug, its active ingredients are not immediately available in the body. Medicines given orally, for example, must first pass through the gastrointestinal tract, where they are absorbed by the intestinal walls and only then absorbed into the blood and passed on to the liver. The time it takes for the substance to reach the plasma and be transported via the bloodstream to the intended location corresponds to its bioavailability.
The bioavailability is therefore a measurable parameter and is often officially stated on drugs. To measure the size, for example, after the oral administration of the respective drug or active ingredient, its concentration in the plasma is determined at different time periods. The measurements usually result in a graph with a curve-like profile, which makes the inundation of the administered agent or active ingredient visible. What is under the curve is called AUC and corresponds to a closed "area under the curve". This area shows a proportional behavior to the respective amount of active ingredients that have reached the organism with the administration. Formulas are available to calculate the absolute bioavailability. The formula F = AUC (peroral) / AUC (intravenous) gives the absolute value.
In the case of pharmaceuticals, the size of the bioavailability is decisive for determining bioequivalence. Bioequivalence is always used when two medicinal products have the same active ingredients and are at the same time interchangeable with one another, although they differ from one another in the manufacturing process or in their auxiliary substances. If both drugs have the same active ingredient but have different bioavailability, they are not bioequivalent and therefore cannot be interchanged with one another.
So-called bioenhancers are available to the pharmaceutical industry to influence bioavailability. They increase bioavailability by increasing the absorption of certain substances in the intestine. In addition, they inhibit the breakdown of substances within the liver and improve the ability of the active ingredients to bind to the intended binding sites. In addition, some bioenhancers increase the possibility that active ingredients will cross the blood-brain barrier.
Illnesses & ailments
Under certain circumstances, the bioavailability of certain active substances or drugs can be reduced. For example, the drugs and active ingredients can break down when the drug passes through the liver for the first time when administered orally. This effect is known as the first pass effect. After absorption, the active ingredient reaches the liver via the portal vein. There it is partially metabolized by the cells of the liver. In this way, only some of the active ingredients actually contained reach the inferior vena cava. This means that only the remaining part of the drug can be used for systemic distribution.
The first-pass effect is usually circumvented by administering the drug parenteral, sublingual, rectal or buccal. Another possibility is the administration of so-called prodrugs. These drugs contain inactive or at least slightly active substances that only become active after being metabolized by the liver. Prodrugs are always of great importance when an actually active active ingredient does not arrive at the desired site of action at all, is reduced or is insufficiently selective when administered orally. The prodrug concept improves the pharmacokinetic properties of the active ingredients and, with oral absorption, also improves the bioavailability of the drugs by reducing the first-pass effect or enabling certain drugs to cross the blood-brain barrier.
The bioavailability of a drug can differ from person to person. For example, the proportion of systemically distributed active ingredients in each drug depends heavily on the respective function of the liver and is not only influenced by the chemical properties of the drug. For example, bioavailability increases automatically in people with certain liver diseases. The same applies to older people, whose liver is only functional to a limited extent for physiological reasons.
In patients with liver disease, the standard dosage of a certain drug can lead to dangerous concentrations of the active ingredients in the plasma and thus develop undesirable effects. Knowing about the liver values of patients is therefore one of the most important bases for deciding on a specific drug therapy or drug management.







.jpg)

.jpg)


.jpg)