Chemokines are small signal proteins that trigger the chemotaxis (migration) of cells. Usually these cells are immune cells. Thus, the chemokines are responsible for the effective functioning of the immune system.
What are chemokines?
Chemokines are small proteins that belong to the cytokine family. They cause cells to migrate. Mainly these are immune cells that are supposed to get quickly to the appropriate location of an injury or infection.
The chemokines are produced by the cells, which they are supposed to attract. On the surface of these cells are receptors that enable the chemokines to dock. The signaling molecules are divided into inflammatory and homeostatic chemokines. In most cases, the chemokines are inflammatory. They attract immune cells to their destination, which immediately trigger inflammatory processes there to defend themselves against infection. Inflammatory chemokines are always produced at the site of the injury or infection by the immune cells present there in order to attract further defense cells.
Homeostatic chemokines are constantly being produced even when there is no infection. They are used to monitor healthy tissue. Chemokines have a chemotaxic effect on immune cells such as monocytes, macrophages, keratinocytes, fibroblasts, platelets, endothelial cells, T cells, stoma cells, neutrophils, and dendritic cells. They are also produced by these cells as signaling substances in order to attract similar cells if necessary.
Anatomy & structure
Chemokines are small protein chains with 75 to 125 amino acids each. At the terminal end of the chain there are one or two cysteine residues. Cysteine is a sulfur-containing amino acid that can form disulfide bridges in the molecule. The cysteine residues now form sulfide bridges within the protein chain.
While the amino acid sequence is variable within the proteins of the chemokine family, the tertiary structure remains the same for all chemokines. The main body is formed as a three-strand anti-parallel sheet with a beta structure. The chain ends at the carboxy terminus with an alpha helix. There are now the cysteine residues. There are four structures in which these terminal cysteine residues can be arranged. Each structure symbolizes a family of chemokines. So two cysteine residues can follow one another. The corresponding chemokine family is called the CC family. If another amino acid is switched between the cysteine residues, it is the CXC family. The CX3C family contains two cysteine residues separated by three amino acids.
Finally, there is a family with a cysteine residue known as the C family. All cysteine residues form a sulfide bridge within the chain. The individual chemokine families have different functions. The exact structure of the chemokines is still not fully understood. The chemokines do not necessarily need tissue fluid or blood to perform their function. They can also pass on their signals via solid structures through concentration gradients. With the positive charge of their many basic amino acids, they bind to a negatively charged sugar molecule (glycosaminoglucan) on the surface of cells. It is not yet clear why they lose their function when they can no longer bind to glycosaminoglucan.
Function & tasks
The main function of the chemokines is to attract certain immune cells to the places in the body that are currently subject to a higher level of defense against infectious intruders. This makes the immune response more effective. In most cases, they also ensure that considerable inflammatory reactions develop in order to ward off the infection. They are generated at the site of the injury or infection by the immune cells already present there.
The now attracted cells move towards the highest concentration of the chemokines. The corresponding chemokine receptors are located on their surface. The chemokines bind to these receptors and thus trigger a migration of the cells towards the highest chemokine concentration. However, each chemokine family binds to its own receptors.The CC family ensures the migration of monocytes, lymphocytes as well as basophils and eosinophils. The CXC family is responsible for angiogenesis (growth of blood vessels). The CX3C family plays a role in inflammatory processes in the nervous system. Finally, the C chemokines activate the CD8 T cells and NK cells (natural killer cells).
You can find your medication here
➔ Medicines to strengthen the defense and immune systemDiseases
If the interaction of chemokines and chemokine receptors is disrupted, the immune system malfunctions. Often, due to a mutation in the corresponding receptor, it is no longer a perfect fit for the docking of chemokines. This means that immune cells can no longer be attracted in critical situations.
This malfunction then manifests itself as an immune deficiency. The so-called WHIM syndrome, a special immune deficiency, can be traced back to a chemokine receptor defect. This disease manifests itself in recurring viral and bacterial infections. The patients show a particular susceptibility to the human papillomavirus, the infection of which is expressed as warts. The bone marrow is full of T precursor cells, but these do not migrate to the sites of infection. Selective immune deficiencies against certain pathogens are also possible. For example, if a receptor for a chemokine of the CC family is mutated, there is a special susceptibility to the West Nile virus. In the event of a mutation, the same receptor also ensures hereditary immunity to the HI virus.
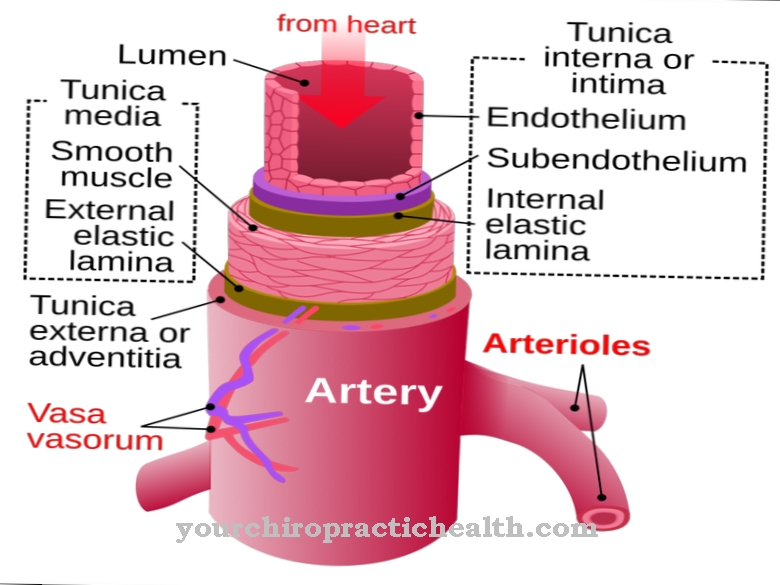
Certain mutations in the area of chemokine receptors can also be partly responsible for autoimmune diseases or allergies. The overproduction of certain chemokines can also lead to illness. It was found that the development of psoriasis (psoriasis) is related to an overproduction of the CXC chemokine IL-8. Rheumatoid arthritis also occurs along with an overproduction of IL-8. Atherosclerotic changes are often the result of excessive inflammatory processes, which are sometimes caused by increased chemokine activities.





.jpg)
.jpg)
.jpg)



.jpg)