Glycerin belongs to the sugar alcohols and is involved in numerous metabolic processes. Medicine uses it, among other things, for the therapy of brain edema, as a laxative in suppositories and, on an experimental basis, to stabilize some operations.
What is glycerin?
Glycerin is an alcohol. Carl Wilhelm Scheele discovered the substance as early as 1779 when he received it when making soap from olive oil. But only in the next century, in 1813, did Michel-Eugène Chevreul finally prove that glycerine is a component of fats - alongside fatty acids. However, it was another ten years before glycerin got its name.
Today the substance is also under the names Glycerol, Propanetriol, Propane-1,2,3-triol or 1,2,3 propanetriol known. The E number for glycerine is 422. In its pure form, it is colorless and liquid; it has a melting point of 18 ° C and a sweet taste. Heating causes glycerin to evaporate. In doing so, it turns into propenal.
Function, effect & tasks
Glycerine is used in various biological processes or can intervene in the course of the processes. It can be found primarily in metabolic processes.
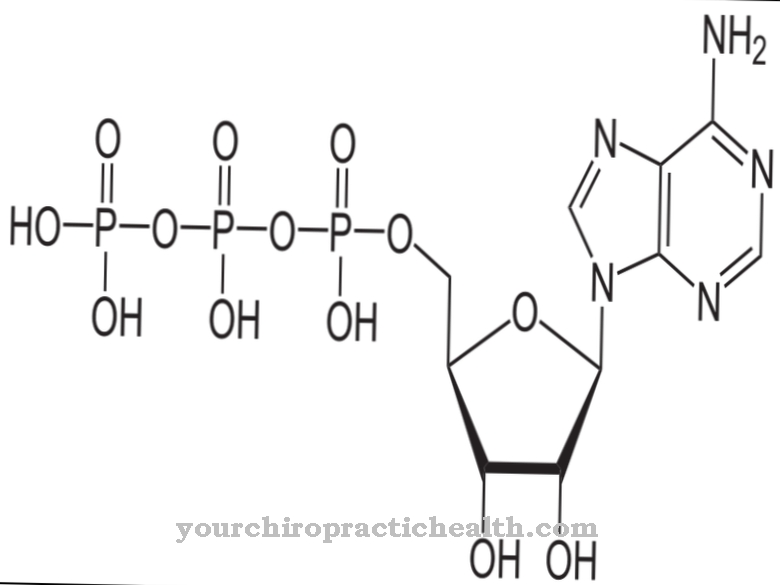
The digestion of fats releases glycerin in the intestines. The tissue reabsorbs the substance and it eventually reaches the liver. The enzyme glycerine kinase converts the glycerine into glycerine-3-phosphate. In addition to the substrate, the enzyme also needs adenosine triphosphate (ATP) for this process. The breakdown of ATP releases energy which glycerol kinase uses to catalyze the biochemical reaction with glycerol-3-phosphate. The body does not always convert the ingested glycerine into glycerine-3-phosphate. Alternatively, the substance can also oxidize and phosphorylate - the product is then glyceraldehyde-3-phosphate or glyceric acid-2-phosphate.
In this form, it intervenes in the metabolism and plays a role in the processing of fats and carbohydrates, for example. The body can use glyceraldehyde in glycolysis. Glycolysis is the process by which cells break down the sugar glucose in a specific way. Glycolysis follows the Embden-Meyerhof scheme, which includes other molecules with glycerine.
Glycerine also plays a role in biomembranes such as cell membranes. Phospholipids form the lipid bilayer that makes up the membrane. The content of phospholipids in the membrane depends on the type of cell; Schwann cells, for example, have a very high content. Phospholipids are made up of fatty acids and phosphoric acids that are esterified with an alcohol, among other things. In addition to glycerine, sphingosine can also be used for this purpose.
Education, occurrence, properties & optimal values
Glycerine occurs mainly in fats and fatty acid esters. Biochemistry also calls the latter triglycerides, as they are triple esters of glycerine. The concentration of triglycerides in the blood is an indicator of the person's health. A value above 150 mg per dl is considered to be elevated and can, for example, be a sign of a lipid metabolism disorder (hypertriglyceridaemia).
Glycerine is made up of carbon, hydrogen and oxygen and has the empirical formula C3H8O3. It is the simplest trivalent alcohol. Glycerine often appears in association with carboxylic acids, which are organic compounds and have at least one carboxyl group. Together with carboxylic acids, glycerine forms organic esters, which in many molecules form compounds with other building blocks.
Diseases & Disorders
An increase in triglycerides in the blood can indicate a lipid metabolism disorder. Medicine calls this disease hypertriglyceridemia. There are various possible causes for the disease. A variant of hypertriglyceridemia is genetic. Affected people suffer from a lack of certain enzymes. Lipoprotein lipase is one such enzyme.
It converts the triglycerides and water into diacylglycerol and fatty acid. Body cells need the fatty acids in order to synthesize fats and store them as reserves, for example. A mutation leads to errors in the formation of the enzyme lipoprotein lipase, which disrupts fat synthesis. In this case, a change in the LPL gene is responsible for the hypertriglyceridemia.
Another cause of the disease can be found in apolipoprotein C2. It is part of the lipoproteins that help transport lipids through the blood. As a result of a mutation in the corresponding DNA segment, not only can hypertriglyceridemia occur; the disturbed lipid metabolism also increases the risk of hardening of the arteries, which medicine calls arteriosclerosis. Atherosclerosis can be due to blood lipids such as triglycerides, which are deposited in the blood vessels and thereby narrow the veins. Complete vessel occlusions are also possible.
The deposits can also loosen and clog fine veins. Possible consequences are stroke, heart attack and pulmonary embolism. Lifestyle factors such as poor diet, tobacco use, lack of exercise and high blood pressure further increase the risk of such complications. Glycerine is also used as a base for ointments and creams. It is also used as a drug: in suppositories, glycerine has a laxative effect and it drives off gallstones and urinary stones. Doctors also use glycerine to treat cerebral edema (water retention in the brain).
Furthermore, there are first attempts to use glycerine during long operations: Doctors could use this to further reduce the body temperature during the operation, which may increase the probability of the intervention being successful. Orally administered glycerin in larger amounts is potentially harmful to health if it is not taken for medical reasons and without medical supervision.




.jpg)

.jpg)


.jpg)