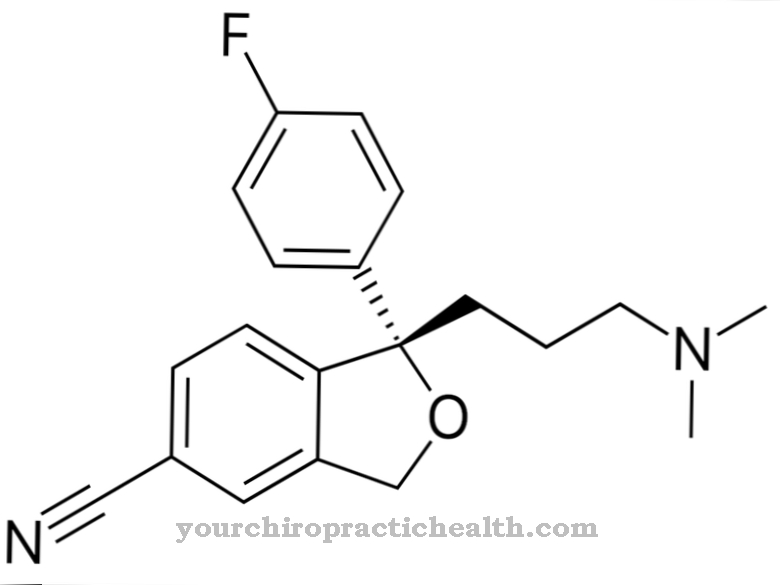
Primidon is an anticonvulsant from the pharmacologically active group of antispasmodic drugs. It is used for long-term therapy of various forms of epilepsy.
What is primidon?

Primidone has an anticonvulsant effect in patients with epilepsy. It is assigned to the active ingredient group of anti-epileptics. The chemical classification takes place in the group of barbiturates.
It is a prodrug, a precursor of the medically active substance. The human organism converts primidone into the powerful phenobarbital (deoxyphenobarbital), which dissolves epilepsy spasms. It is a breakdown substance (metabolite). This drug is administered by doctors in special forms of epilepsy.
Pharmacological effect
The medical mechanism has not yet been fully explored. However, medical professionals assume that primidone affects the membrane (cell wall) of the nerve cells and thus has an anti-seizure effect.
Primidone is known from the long-term therapy of various forms of epilepsy. These special forms include grand mal epilepsy, petit mal epilepsy, status epilepticus, and myoclonic seizures. In children with petit mal epilepsy, Primidon has a preventive effect with regard to the development of grand mal epilepsy. These diseases appear as temporal lobe seizures, as well as primarily generalized seizures that affect the whole brain (grand mal epilepsy).
Secondary generalized seizures, which affect individual areas of the brain (petit mal epilepsy), form another group. Gran mal seizures are also known as tonic-clonic seizures. The tonic phase lasts about 10 to 30 seconds and is characterized by muscle tension and cramps. During the clonic phase, muscle tremors and irregular convulsions of great intensity occur. This phase can last anywhere from thirty seconds to three minutes. Symptoms include a short, intense scream immediately before the onset of convulsions, salivation, twitching, wetting, twisting of the eyes, sudden end of convulsions, unconsciousness and then increased feeling of tiredness.
Since it is not possible to predict the occurrence of seizures exactly, only preventive or soothing treatment with suitable drugs such as primidone remains. This drug is the second choice for anesthesia preparation and treatment of essential tremor when the first choice drugs have been shown to be ineffective.
Medical application & use
After the substance has been absorbed by the organism, it is immediately metabolized or converted into phenobarbital. Another active ingredient that develops through this process is phenylethylmalonamide, which, however, is of little importance.
Primidon works together with the breakdown product phenobarbital at the central switching point of the neurotransmitter gamma-aminobutyric acid (GABA). Besides glutamate, it is the most important inhibitory neurotransmitter of the central nervous system. It plays an important role in many neuronal processes in the CNS. It has a modulating effect and acts as an opponent (antagonistic) to the excitatory (exciting) acting glutamate. Phenobarbital has a strengthening effect on the concentration of the messenger substance gamma-aminobutyric acid and reduces the susceptibility to seizures.
Risks & side effects
Primidon also has side effects and drug interactions. The drug must not be administered in the case of known hypersensitivity to the drug substance or to other barbiturates. Central depressant drugs such as antidepressants, sleeping pills, opioid pain relievers and neuroleptics are contraindications, as their mode of action is enhanced by primidone.
Simultaneous use of the calcium blocker nimodepine for the treatment of angina pectoris (tight chest), hypertension (high blood pressure) and tachycardia (rapid heartbeat) can be fatal. This drug is not given to patients with signs of acute alcohol intoxication.
A careful risk-benefit assessment must be made in patients with cardiac arrhythmias, diseases of the respiratory system, severe sepsis and functional disorders of the liver and kidneys. The reason for a possible contraindication lies in the considerable delay in the breakdown of the substance in the event of previous damage to the organism. In most cases, health professionals will use a reduced dose of primidone while carefully monitoring the patient.
The barbiturate may only be administered in very small doses during pregnancy and breastfeeding if treatment is absolutely necessary. Common side effects are nausea, vomiting, headache, dizziness, increased excitability, memory and language disorders, incoordination, impaired perception, indigestion, convulsions, tremors, drowsiness and delayed reaction times.
Cardiac arrhythmias, pigment disorders, blood formation disorders, anemia, skin changes, allergies, muscle fatigue and liver dysfunction occur rarely.
Long-term therapy increases the risk of developing osteoporotic diseases. In the elderly and children, increased excitability, aggressiveness and moodiness were often observed. The breakdown of beta-blockers used to treat heart conditions and high blood pressure is accelerated while the effect is decreased.
Primidon reduces the effect of cardiac glycosides such as digitoxin and cytostatics used to treat tumor diseases. The effect of anti-epileptic drugs such as diazepam, clonazepam, phenytoin, carbamazepine is reduced.
Side effects and toxicity of methotrexate used to treat cancer are exacerbated. Regular checks of liver enzyme values and blood counts are particularly indicated in the initial phase. Patients with previous illnesses who are taking [cortisone]] at the same time have an increased risk of osteoporosis.
Primidone can affect the way contraceptives work, so additional contraception should be used.
Experts believe that taking the drug increases the risk of suicide, so regular patient monitoring is essential. As with most medications, there is also the possibility of a habituation effect with primidone. To avoid withdrawal symptoms, the drug must not be stopped suddenly, but must be reduced gradually. With cold withdrawal, brain seizures are possible.
Due to the unfavorable side effect profile, Primidone is a second choice drug. It is only used if no alternative drugs with a more favorable appearance are available or the drugs of first choice have proven to be ineffective.











-eisenmangelanmie.jpg)
.jpg)