At the Salvage Pathway a new biomolecule is synthesized from the degradation products of a biomolecule. The salvage pathway is also known as the recovery path and is, so to speak, a form of recycling within the metabolism.
What is the salvage pathway?
Salvage pathway denotes on the one hand the general form of this recycling within the metabolism and on the other hand the metabolic pathway of the purine nucleotides. Purine nucleotides are the basic chemical building blocks of deoxyribonucleic acid (DNA) and ribonucleic acid (RNA).
In putin nucleotide salvage, mononucleotides are formed from the purine bases guanine, adenine and hypoxanthine. At 90%, this metabolic pathway is the main metabolic pathway for free purines. The rest is broken down into uric acid. The salvage pathway offers numerous advantages, especially compared to the de novo biosynthesis of purine mononucleotides. For example, it is significantly more energy efficient.
Anatomy & structure
The synthesis of the bicyclic purine bases involves a lot of effort for the body. Therefore, they are broken down into simple bases and then used again.
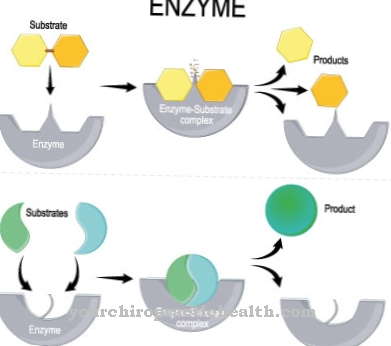
In the recycling pathway, various intermediate products of the breakdown of mononucleotides, nucleosides, polynucleotides or nucleic acid bases are used in building reactions instead of breaking them down completely. Through the salvage pathway reaction, useful and valuable intermediate products from the metabolism, so-called metabolites, can be saved from disposal. So these metabolites do not have to be produced again. This process saves the cell high energy consumption. In the salvage pathway, a ribose phosphate from phosphoribosyl pyrophosphate (PRPP) is transferred to a free purine base.
The nucleotide is formed by splitting off pyrophosphate. The enzymes required for this are activated by the phosphoribosyl pyrophosphate and inhibited by the end products. From the purine base adenine, adenosine monophosphate (AMP) is formed together with (PRPP) and by means of the enzyme adenine phosphoribosyl transferase (APRT). In connection with PRPP and the enzyme hypoxanthine-guanine-phosphoribosyltransferase (HGPRT), guanine becomes the nucleotide guanosine monophosphate (GMP). With PRPP and the enzyme hypoxanthine-guanine-phosphoribosyltransferase, hypoxanthine becomes the nucleotide inosine monophosphate (IMP).
Other enzymes that are involved in the salvage pathway are nucleoside phosphorylases, nucleoside kinases and nucleotide kinases. 90% of the purines are first converted into nucleotides and then made usable again for the synthesis of nucleic acids through conversions. 10% of the purines are broken down into uric acid and excreted via the kidneys.
Function & tasks
The salvage pathway takes place in almost all cells in the body, as purines are broken down in almost all cells in the body. Purines belong to the group of heterocycles and, alongside pyrimidines, are the most important building blocks of nucleic acids. Purines are formed using the salvage pathway itself. They are contained in all cells that have a nucleus.
Food of animal origin, especially offal and skin, contain a lot of purines. The purines that are not recycled via the salvage pathway are broken down into uric acid and excreted through the kidneys. There are no blood values for the salvage pathway, but there are for uric acid. In men, uric acid levels in the blood are usually between 3.4 and 7.0 mg / 100 ml. In women, the uric acid value should be between 2.4 and 5.7 mg / l.
Diseases
If there is a defect in the salvage pathway, the purines can no longer be recycled. Significantly more purines are broken down, so that more uric acid is produced. The kidneys are no longer able to completely excrete uric acid, leading to hyperuricemia.
Hyperuricemia is an increase in the level of uric acid in the blood. By definition, hyperuricaemia is present from a uric acid level of 6.5 mg / dl. The limit value applies equally to both sexes. An increase in uric acid levels due to a disruption of the salvage pathway is also known as primary hyperuricemia. About 1% of all hyperuricemia is caused by an overproduction of uric acid due to a disturbance in the purine metabolism. The majority of primary hyperuricemia is based on decreased uric acid excretion in the kidneys.
In order to distinguish whether the increased urine values are based on reduced excretion or on increased uric acid production, the uric acid clearance must be determined. To calculate the uric acid clearance, the uric acid excretion in the 24-hour urine collection and the serum uric acid are determined.
In most cases, hyperuricemia remains asymptomatic. In the case of massive hyperuricemia, an acute attack of gout occurs. This is where the crystallized salts of uric acid are deposited in the joints. This leads to inflammation in the affected joints with overheating, pain and severe redness. The metatarsophalangeal joint, the ankle joint and the knee joint are particularly often affected. If the gout persists for a long time, the tissue is remodeled. The cartilage in the joints thickens and so-called gouty tophi develop.
A genetic defect that leads to hyperuricemia is Lesch-Nyhan syndrome. The disease is inherited in an X-linked recessive manner and results in a deficiency in the enzyme hypoxanthine-guanine-phosphoribosyltransferase (HGPRT). Since the enzyme is involved in the purine metabolism of the purine bases hypoxanthine and guanine, more purines are produced for degradation. The result is a sharp rise in uric acid. The disease is inherited in an X-linked manner. That is why almost exclusively men are affected by Lesch-Nyhan syndrome. The first symptoms appear around ten months after the birth.
The children show a conspicuous stance in combination with a sedentary lifestyle and developmental deficits. The first sign is often an increased urine residue in the diaper. In severe cases, self-injuries such as lip and finger bites and impaired thinking can also occur. The affected children may also behave aggressively towards their parents, siblings, friends or carers.











.jpg)