At Sorivudine is a medical drug that was developed in Japan to treat herpes. Sorivudine was sold under the trade name Usevir® and has not been available since a drug scandal that killed several people in Japan. In Europe there was no approval at all, so the preparation did not have to be withdrawn from the market.
What is sorivudine?
Sorivudine was developed by the Japanese pharmaceutical company Nippon Shoji in the early 1990s. The active ingredient was used to treat herpes infections and acted as a virostat. These are drugs that inhibit the replication of viruses and thereby enable the treatment of infectious diseases.
Sorivudine is effective against infections caused by herpes simplex type 1 viruses. There was also an indication for Epstein-Barr viruses (often also called EBV or HHV4).
Sorivudine is described in chemistry and pharmacology by the empirical formula C 11 - H 13 - Br - N 2 - O 6 and has a moral mass of 349.13 g / mol. After Usevir®, the most important sorivudine-containing preparation, triggered a national drug scandal in 1994, the substance was withdrawn from the market. Since then it has not been available as a medicinal product anywhere in the world and is not used in human medicine.
Pharmacological effect
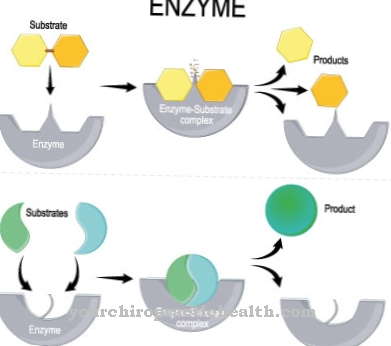
Sorivudine is effective against Herpes and Epstein-Barr viruses by inhibiting the enzyme dihydropyrimidine dehydrogenase (DPD). This is u. a. responsible for the breakdown of various pyrimidines and fluorouracil. Fluorouracil, often also called 5-FU or 5-fluorouracil, is a cytostatic agent. As such, the substance is administered as part of extensive chemotherapy to treat various cancers.
Due to the inhibition of DPD caused by sorivudine, the degradation of fluorouracil is made impossible or slowed down considerably, which can be fatal under certain circumstances. So there are massive interactions between sorivudine and fluorouracil.
Sorivudine itself is broken down in the body by bromovinyluracil. The breakdown process largely corresponds to that of the virostat brivudine, which is still approved as a drug.
Medical application & use
Sorivudine was used and manufactured to treat herpes. The preparation Usevir® was indicated to treat infections with herpes zoster (shingles) or herpes simplex type 1. Epstein-Barr viruses could also be treated with sorivudine. It was taken exclusively orally in the form of film-coated tablets. Preparations containing sorivudine were subject to pharmacy and prescription requirements in Japan.
Sorivudine was withdrawn from the market after a drug scandal in Japan. Due to the way the drug works, a total of 16 patients died in 1994 after taking sorivudine because they were previously treated with fluorouracil. This was possible because Nippon Shoji, the manufacturer of the sorivudine, provided inaccurate information to the responsible Ministry of Health during the approval process. Because even while the drug tests required in advance were being carried out, there were deaths caused by the known interaction of fluorouracil and sorivudine. Since the manufacturer failed to pass on the necessary information after the end of the test phase, sorivudine was approved. There was no indication of the lethal interaction, so that numerous co-administrations with fluorouracil occurred.
Risks & side effects
Sorivudine must not be taken if there are known intolerances or allergies to the agent. These can manifest themselves in massive skin reactions such as itching, redness or rashes. The drug can also cause headaches and general malaise as an undesirable side effect. Symptoms of the gastrointestinal tract are also conceivable.
Sorivudine leads to massive interactions with fluorouracil, since the breakdown of the cancer drug is inhibited by sorivudine. This can be fatal under certain circumstances. From a medical point of view, this is a contraindication, so that combined administration of fluorouracil and sorivudine must absolutely be avoided.












.jpg)