G-CSF is a peptide hormone that stimulates the formation of granulocytes. It is therefore of great importance for the function of the immune system. The hormone is also given as a drug to patients with a severely weakened immune system to stimulate the production of neutrophils.
What is G-CSF?
G-CSF is the abbreviation for the name Granulocyte Colony Stimulating Factor. It is a peptide hormone that stimulates the formation of granulocytes from pluripotent stem cells. The granulocyte colony-stimulating factor belongs to the cytokines.
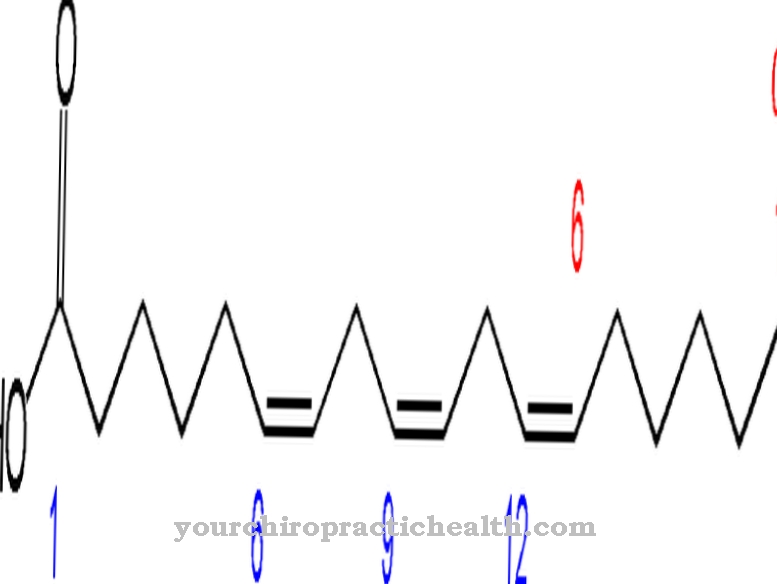
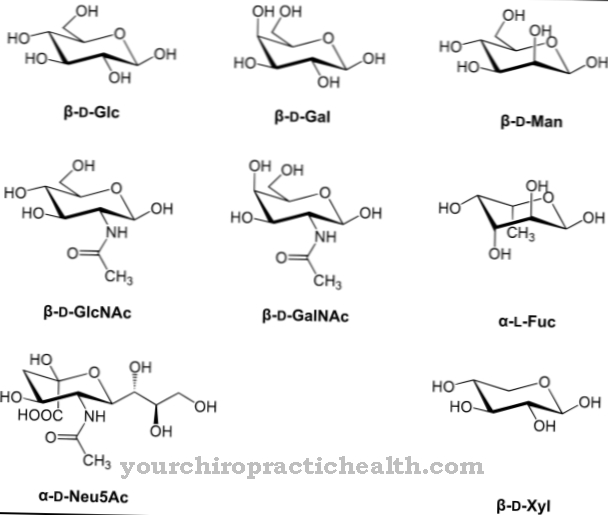
In general, cytokines are proteins that are responsible for the proliferation of immune cells and thus control the immune response. There are different types of cytokines. The peptide hormone G-CSF is one of the colony-stimulating factors. In chemical terms, human G-CSF is a glycoprotein made up of 174 amino acids. At 133 there is the amino acid threonine, which is glycosylated at its hydroxyl group. The non-proteinogenic portion of the molecule at the glycosylated site is approximately four percent of the molecular weight. It consists of the components α-N-acetyl-neuraminic acid, N-acetyl-galactosamine and β-galactose.
The glycosylation has a stabilizing effect on the protein. At the same time, it also plays an important role in certain functions, such as activating mature granulocytes to combat current sources of infection. G-CSF also contains two disulfide bridges, which determine the secondary structure of the protein. The coding gene for G-CSF is located on chromosome 17 in humans.
Function, effect & tasks
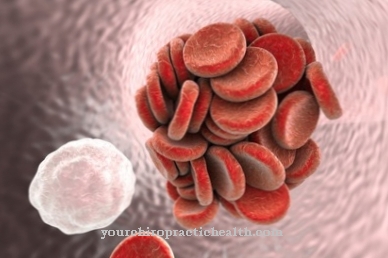
As mentioned earlier, G-CSF is an important factor in the immune system. It stimulates the immature progenitor cells of the blood-forming system (haematopoietic system or pre-CFU) to differentiate and proliferate. This means that the undifferentiated pluripotent stem cells under the influence of G-CSF differentiate into granulocytes and multiply through cell division.
Granulocytes are neutrophilic white blood cells that act as so-called phagocytes. These become effective when the organism is infected by bacteria. With every bacterial infection, phagocytes multiply from undifferentiated progenitor cells. G-CSF also stimulates mature granulocytes to move to the sources of infection in order to kill the bacteria there. In this function, the molecule is supported by its glycosylation-bound residue. At the source of the infection, G-CSF can thus increase the formation of hydrogen peroxide in the granulocytes, which makes the killing of bacteria even more effective.
A third function of G-CSF is to cause the hematopoietic progenitor cells to detach from their environment in the bone marrow. As a result, some of these cells get into the peripheral blood. With the help of further doses of G-CSF, this process can be repeated, whereby pluripotent stem cells accumulate in the blood. This process is also known as apheresis. Apheresis has proven useful for stem cell donors or for patients exposed to intense chemotherapy. In this way, chemotherapy patients can have their own blood enriched with stem cells transplanted again.
Stem cell donors, on the other hand, can make a normal blood donation instead of a bone marrow donation. G-CSF serves as a medicinal substance and is used in chronic neutropenia (reduction in neutrophil granulocytes), in chemotherapy or in stem cell transplants.
Education, occurrence, properties & optimal values
G-CSF is integrated into the complex homeostatic network of the organism. The granulocyte colony-stimulating factor is part of both the immune system and the endocrine system. The pluripotent stem cells of the bone marrow and the mature neutrophil granulocytes have receptors for G-CSF.
When necessary, the proteins of G-CSF bind to the receptors and thus ensure that their effect unfolds. Every organism forms its own G-CSF. However, if the need increases, such as with severe infections, chemotherapy or general immunodeficiency, the hormone may have to be injected subcutaneously. Well-known drugs are pegfilgrastim and lipegfilgrastim. These are produced recombinantly from certain mammalian cells such as CHO cells (Chinese Hamster Ovary) or from Escherichia coli. The amino acid sequences are identical in both production forms.
There may be differences in glycosylation. However, newer products are glycosylated in the same position as the original G-CSF. Certain forms of processing such as PEGylation increase the resistance and half-life of the drugs when used without changing their effectiveness. For this purpose, G-CSF is chemically bonded to polyethylene glycol.
Diseases & Disorders
There can also be side effects when using G-CSF. Bone and muscle pain are the most common. This is often accompanied by nausea, vomiting, loss of appetite and diarrhea. Inflammation of the mucous membranes and hair loss can also occur. The complaints are the result of the increased formation of neutrophils, which then trigger increased immune reactions.
Infiltrates in the lungs, which among other things cause coughing, shortness of breath and fever, are observed less frequently. This can even lead to the so-called Acute Respiratory Distress Syndrome (ARDS), which indicates an intensive reaction of the lungs to external damaging factors. The spleen can enlarge so much that it ruptures. Another symptom is increased leukocytosis, i.e. the increased formation of white blood cells. In the presence of sickle cell anemia, G-CSF must not be used because, according to an American study, severe side effects can occur here, some of which even lead to multiple organ failure.
However, many studies also show that the symptoms are usually reversible. After discontinuing therapy with G-CSF, the side effects also disappear. Although there is an increased formation of neutrophils during treatment with G-CSF, studies have so far not found any increased risk of developing leukemia.



.jpg)