The catalysis corresponds to the reduction in activation energy required for chemical and biological reactions. The catalytic reduction in the amount of energy required is made possible by a catalyst that corresponds to an enzyme in biology. In enzymatic diseases, the catalytic properties of enzymes can be reduced or even abolished.
What is catalysis?

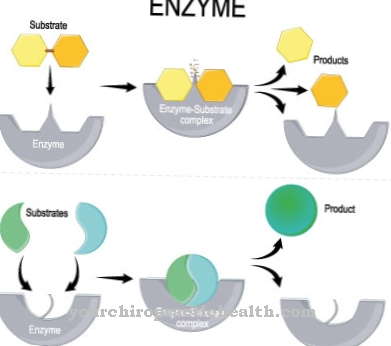
Enzymes have special tasks in the human body. As different as the function of individual enzymes may be, they basically all have the same task and have similar properties to fulfill this task. The main task of all enzymes is catalysis. Therefore, biochemistry says they have catalytic properties.
Literally translated, catalysis means "dissolution". The focus of catalysis is the activation energy. As such, chemistry describes the amount of energy that is absolutely necessary in a reaction system for the chemical reaction of both reaction partners. Catalysts are used to reduce the activation energy and thus allow both reactants to react in the reaction system even at low energies. In biological reaction systems, enzymes with catalytic properties reduce the activation energy of a certain chemical reaction and accordingly function as chemical catalysts.
In the context of a catalysis, on the one hand, the probability of a successful reaction process increases and, on the other hand, the speed of the reaction sometimes increases. A shift in the chemical equilibrium does not occur in the context of catalysis.
Chemistry distinguishes homogeneous catalysis from heterogeneous catalysis. Biocatalysis corresponds neither to one nor the other form. It is an independent form of catalysis.
Function & task
Biocatalysis corresponds to the control, implementation or acceleration of chemical reactions in a biological environment. Enzymes act as biological catalysts in this process. Each enzyme consists largely of proteins, some of which are associated with a co-factor. Almost all biochemical reactions in living organisms have an enzymatic catalyst.
Biocatalysis is implemented in biotechnology using isolated or live enzymes. An example of biocatalysis can be found in beer breweries, where biocatalytic processes are implemented using bacteria, fungi or yeast. The pharmaceutical industry uses biocatalysis to achieve otherwise impracticable reactions.
In the human body, catalysis is constantly taking place, in which enzymes accelerate certain reactions. Enzymes are relevant for the metabolism of organisms, for example, and largely control the biochemical reactions in the metabolic processes. They control digestion, for example, but are also involved in the transcription and replication of DNA in the form of polymerases.
The majority of all biochemical reactions would occur in a living organism without enzymes at a negligibly slow rate. Enzymes accelerate the achievement of chemical equilibrium without changing anything in the equilibrium.
An enzyme has catalytic activity because it can lower the activation energy in chemical reactions. This energy corresponds to the amount of energy that must be applied in advance to initiate a reaction. During the reaction the substrate changes to energetically unfavorable transition states. The activation energy forces the substrate into its transition state. The catalytic effect of enzymes intervenes at this point in the reaction by stabilizing the transition state of the substrate via non-covalent interactions. In this way, significantly less energy is required to convert a substrate into the transition state. Because of this, the substrate converts to the final product of the reaction at a faster rate. With these catalytic functions, the enzymes are considered to be the pioneering elements for every biochemical reaction product.
Illnesses & ailments
If enzymes mutate or no longer perform their catalytic role adequately for other reasons, there are extensive health consequences. The disease group of metabolic diseases includes various disorders from the area of intermediate operating metabolism. Such disorders are either congenital or acquired.
The metabolic diseases vary greatly in their extent and spread. Clinically, too, they manifest themselves very heterogeneously. A related disorder is, for example, the widespread disease diabetes mellitus. However, much rarer hereditary diseases with a lethal course also fall into this group of diseases. Osteopenia and the resulting osteoporosis can also be traced back to metabolic diseases.
Most of the congenital diseases from the superordinate group of metabolic diseases correspond to genetic defects in various enzymes. Depending on the enzyme concerned, its catalytic function and its reaction product, enzymatic defects or enzyme deficiencies can, for example, cause organs to fail.
Gaucher's disease is a relatively rare and inherited metabolic disease. In the context of this disease, the affected enzyme is glucocerebrosidase or glucocerebrosidase. This enzyme breaks down aged components of the cell membrane in a healthy organism. Gaucher's disease is deficient in this important enzyme. If the enzyme does not show sufficient activity, it comes to the deposition of membrane components within the lysosomes. More than 200 mutations of the enzyme have so far been documented in the context of Gaucher's disease. The degree of residual enzymatic activity depends on the mutation of the coding gene in the individual case. The disease can, for example, cause the enzyme to completely fail. However, a functionally weak reduction in enzymatic activity is also conceivable. Most patients with the disease show manifestations of both internal organs and the nervous system.












.jpg)