Progenitor cells have pluripotent properties and form the reservoir in various types of tissue from which somatic tissue cells are formed through proliferation and differentiation.
They arise from the asymmetrical division of pluripotent stem cells, of which one daughter cell develops as a progenitor cell and the other completes the stock of stem cells. Progenitor cells play a crucial role in the formation of new tissue.
What is a progenitor cell?
The term progenitor cells denotes precursor cells of certain types of tissue. They arise through asymmetrical division from adult, multipotent stem cells.
One daughter cell of the divided stem cell develops into a progenitor cell, while the other daughter cell remains in the multipotent stem cell stage and completes the supply of stem cells again. Adult stem cells have so far been detected in over 20 types of tissue. After the division of a stem cell, the progenitor cell loses its multipotency in several steps - stimulated by growth factors - and differentiates into the somatic tissue cell of the tissue for which it is intended.
This means that the original multipotency first changes into a pluripotency, which is necessary for the development of different somatic cells within a tissue, before the cell completely differentiates into somatic tissue cells with complete loss of its multipotency or pluripotency and its ability to divide. There are indications that the increasing determination of progenitor cells on a special tissue is still reversible up to a certain stage of development. The differentiation of the cells is controlled by tissue-specific growth factors.
The research area that deals with progenitor cells is subject to dynamic development, so that no general nomenclature has yet been developed. Some researchers therefore still use the terms progenitor cell and stem cell as synonyms. Because progenitor cells differ from tissue to tissue in terms of their development potential, they are sometimes referred to as determined stem cells.
Anatomy & structure
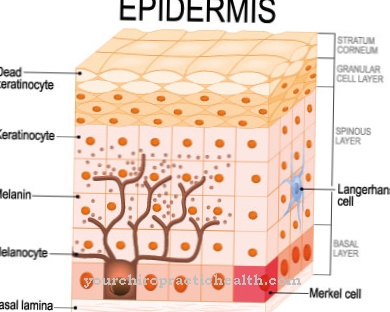
The peculiarity of the progenitor cells is their still partially existing ability to mature into different cells within a tissue. They therefore differ from tissue to tissue. For example, a distinction is made between haematopoietic and endothelial progenitor cells.
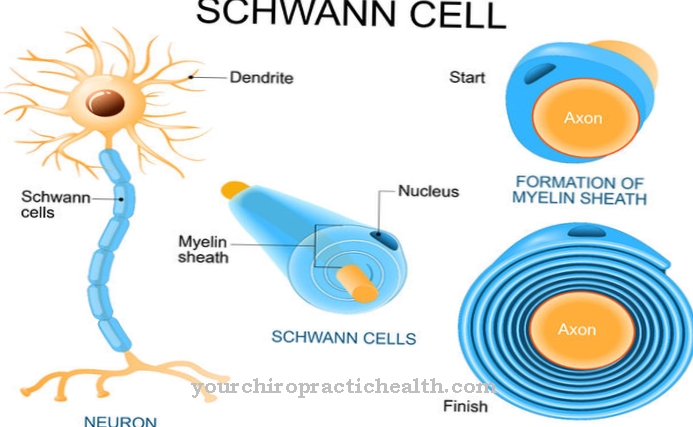
From the hematopoietic precursor cells, which are mainly located in the bone marrow, white or red blood cells can develop in the course of further differentiation steps. Endothelial progenitor cells circulate mainly in the blood and also come from the bone marrow. If necessary, they are used to repair blood vessels and to create new ones (angiogenesis). The endothelial progenitor cells already carry proteins characteristic of vascular endothelia on their surface. To date, progenitor cells have been detected in more than 20 different types of tissue, including in the brain and the peripheral nervous system.
As a rule, progenitor cells specialized in certain types of tissue are called blasts, e.g. B. as osteoblasts, myeloblasts, neuroblasts and many more. They are characterized by the fact that they have usually not yet been finally assigned to a specific type of cell. Typical morphological features of the blasts are an enlarged cell nucleus, a high proportion of endoplasmic reticulum, a high energy metabolism based on a high number of mitochondria and many other features.
Function & tasks
As a rule, differentiated, somatic cells of a certain tissue not only lose their ability to divide, but also their ability to regress into progenitor cells. They are also called unipotent because - if they are still capable of dividing - they can only produce cells of the same type with the same properties when they divide. The loss of the ability to divide differs from tissue type to tissue type and occurs for safety reasons, since otherwise new tissue could constantly form with only minor disturbances, which can almost inevitably lead to problems.
Progenitor cells therefore essentially have the task of replacing tissue cells after injuries or tissue loss caused by disease, or to ensure the necessary replenishment of specialized tissue cells during growth processes. The mobilization of the progenitor cells takes place as required and is controlled by various cytokines and interleukins. Depending on the type of tissue, the progenitor cells act as a patrol in the bloodstream or they represent the hidden reserve for the formation of new tissue cells, which can be mobilized for repair and growth purposes. For example, endothelial progenitor cells play a special role in overcoming sepsis.
Sepsis is usually triggered by bacterial toxins, which lead to increased necrosis and apoptosis (programmed cell death) of endothelial cells in the vessels. It has already been proven that in such cases an increased level of certain cytokines leads to an increased release of endothelial progenitor cells from the bone marrow and thus an increase in the repair mechanism for the restoration of the damaged inner vessel walls.
Diseases
As potential tissue cells, progenitor cells take on the task of stepping in in the event of illness or injury in order to eliminate the tissue damage suffered as well as possible.
The multi-stage activation and differentiation stages of the progenitor cells mean that they can lead to disease symptoms themselves through acquired or genetic, congenital defects. Acute leukemia is a well-known disease of progenitor cells, which are responsible for the replenishment of white or red blood cells or platelets. Malignant progenitor cells spread in the bone marrow and displace the functional progenitor cells. This primarily results in anemia and a lack of blood platelets (thrombocytes).
The malignant cells can spread into almost all tissues, including the skin and mucous membranes. They can even be felt as small nodules in the oral mucosa. Some other cancers are also based on altered stem and progenitor cells. In most cases, these are mutated stem cells, from which correspondingly modified precursor cells develop, which have defects in certain protein complexes and therefore divide unchecked, unaffected by deactivating cytokines.

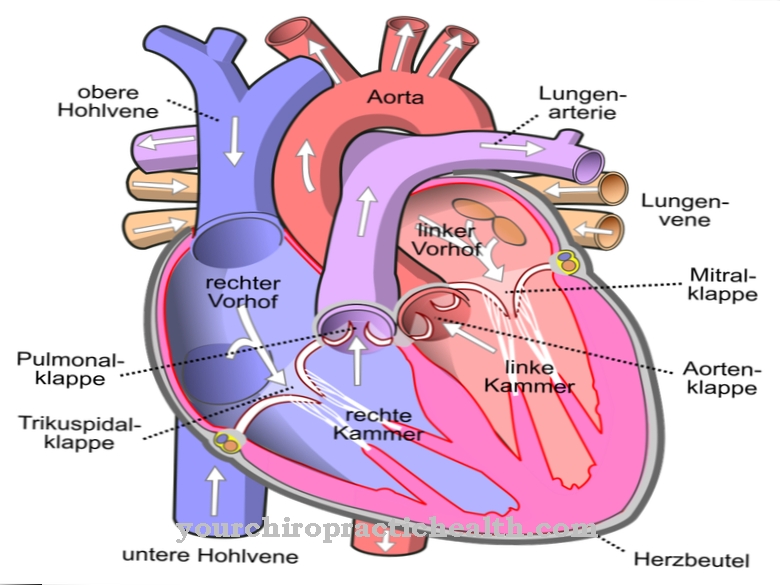




.jpg)